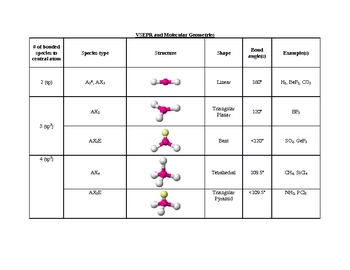
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. Figure 9.2 Shapes of AB2 and AB3 Molecules Figure 9.3 Shapes Allowing Maximum Distances between Atoms in ABn Molecules Table 9.1 Electron-Domain Geometries as a Function of Number of Electron Domains Table 9.2 Electron-Domain and Molecular Geometries for Two, Three, and Four Electron Domains around a Central Atom Table 9.3 Electron-Domain and M. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. TABLE 9. There are five different electron-domain geometries: linear (two electron domains), trigonal planar (three domains), tetrahedral (four domains), trigonal bipyramidal (five domains) and octahedral (six domains). These arrangements are identical to those of the electron-pair geometries predicted by VSEPR theory. The arrangement of electron domains about the central atom of an AB n molecule is its electron-domain geometry. The geometrical arrangements characteristic of the various sets of hybrid orbitals are shown in Figure 5.21. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. (c) Which of the following as- vector in the mole (0) from the carbon atom toward a fluotine atom,(ily frot catbon atom to a point midwary between pects of the VSEPR model is illustrated by part (b) The (if) from the carbon atom to a polnt drogen atoms,or atom (Sections 9.2 and 9.3 electron-domain geometry for four electron rahedral. Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure. The hybridization of an atom is determined based on the number of regions of electron density that surround it. We designate SF 4 as AX 4E it has a total of five electron pairs.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
